
Again, the mass of a mole substance is called the molar mass, and to find the molar mass of a substance you just need a periodic table and the chemical formula. That’s the total molar mass of the compound CO2. Then finally you add all products, so 12 plus 32 is 44. You would multiply it by its subscript of 2, which is 32. Then you’d do the same thing with oxygen. Then the way you would find that is first you would find the atomic mass of carbon, which is 12, and then you’d multiply it by the subscript, which is just an understood 1 right there, so the product there would be 12. If you were trying to find the molar mass of the compound carbon dioxide, which looks like this-CO2-and the molar mass is 44. 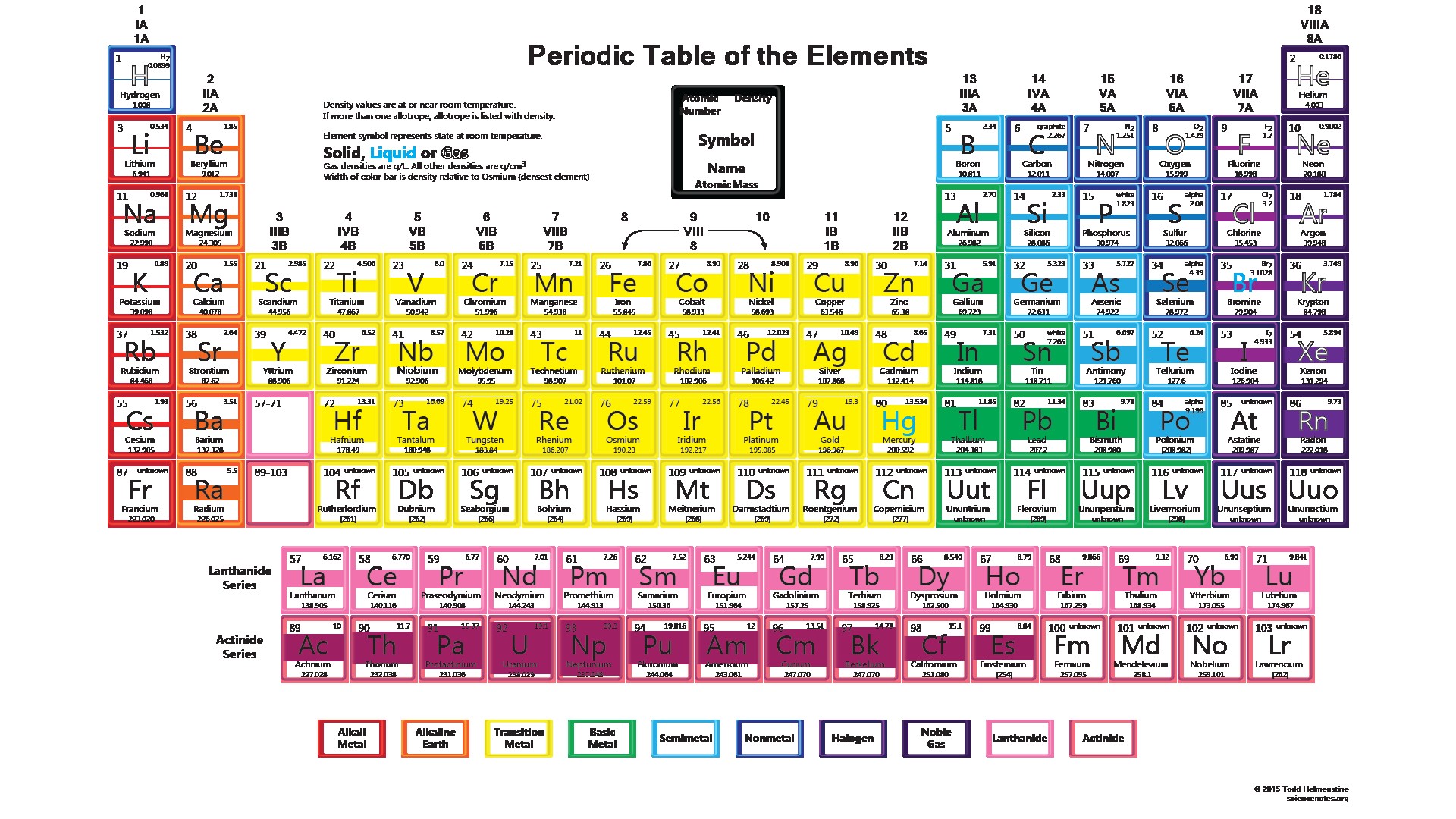
Then, to find the molar mass of the compound, you’re going to add all those products together. Scientists use the periodic table to quickly refer to information about an element, like atomic mass. By multiplying the atomic mass by the subscript you’re getting the product, you’re getting the molar mass product for that atom. The periodic table of chemical elements, often called the periodic table, organizes all discovered chemical elements in rows (called periods) and columns (called groups) according to increasing atomic number. That way, you’re finding the total molar mass for all of that atom because you’re finding the atomic mass, then you’re multiplying it by however many times it appears in the compound. Options for hiding the symbol or name of the elements provide a handy learning aid for memorizing the periodic table. After you’ve found the atomic mass, multiply the atomic mass by the subscript. Use this periodic table for calculating molar mass for any chemical formula. You’re going to start with one atom at a time. I’m going to show you a 3-step process for finding the molar mass of a compound.įirst, find the atomic mass of an atom. You kind of follow the same protocol for finding the molar mass of an element, but then you combine all those molar masses together to find the molar mass of the compound. Now the molar mass of a compound can be calculated by summing the molar masses of each atom and the chemical formula. That’s how you find the molar mass of an element. Now we’ll write it with a different unit because it’s also our molar mass, and that will be grams over moles: \(\frac\). If you’re looking at the element of copper you would look in the periodic table and find that it has an average atomic mass of 63.55 AMU, so that’s the molar mass. If you’re trying to find the molar mass of an element, then you’ll take the average atomic mass listed in the periodic table. To calculate the molar mass of a substance you need a periodic table and the chemical formula. John Alexander Reina Newlands was an English chemist who worked on the development of the periodic table. Despite the fact that Newlands’s table had no logical place for the d-block elements, he was honored for his idea by the Royal Society of London in 1887. At one scientific meeting, Newlands was asked why he didn’t arrange the elements in alphabetical order instead of by atomic mass, since that would make just as much sense! Actually, Newlands was on the right track-with only a few exceptions, atomic mass does increase with atomic number, and similar properties occur every time a set of ns 2 np 6 subshells is filled. Unfortunately, Newlands’s “law of octaves” did not seem to work for elements heavier than calcium, and his idea was publicly ridiculed. There were seven elements because the noble gases were not known at the time. He described octaves as a group of seven elements which correspond to the horizontal rows in the main groups of today's periodic table. Newlands therefore suggested that the elements could be classified into octaves.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
